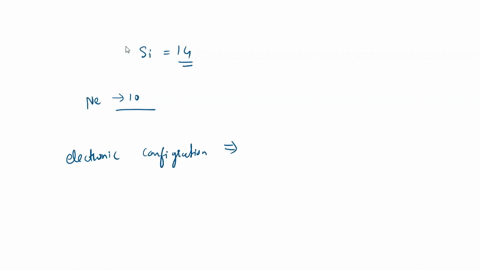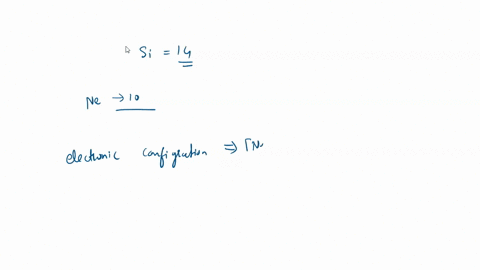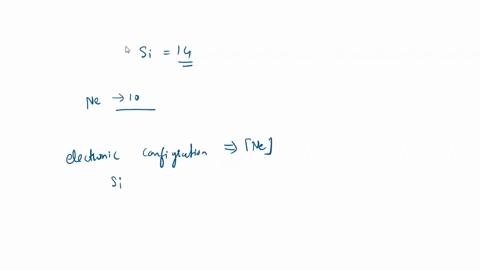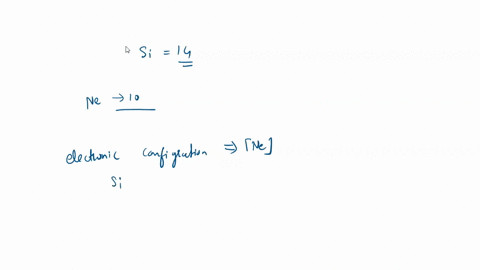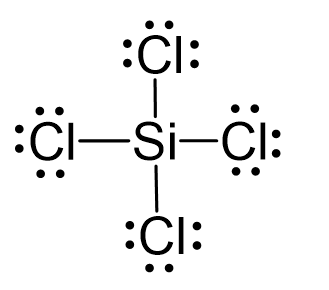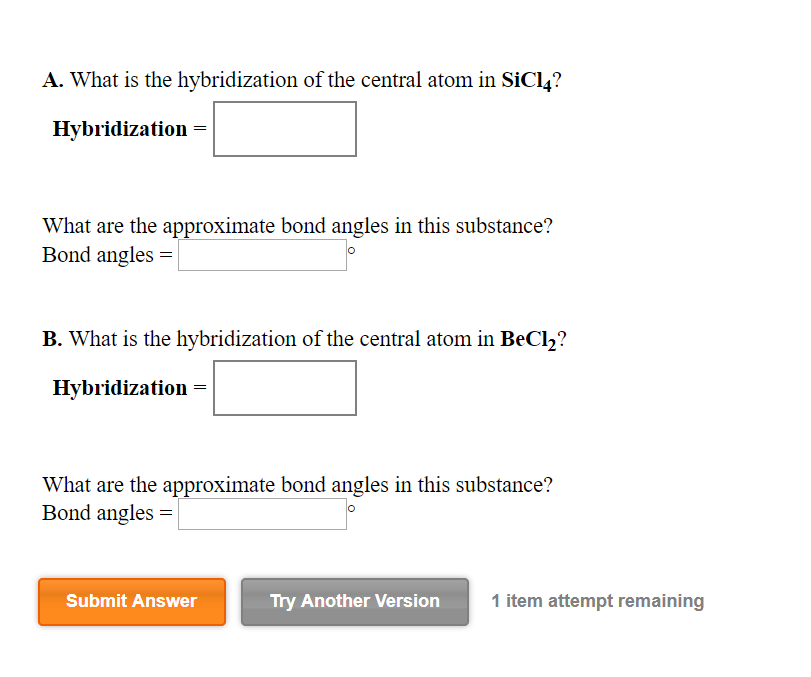
SOLVED: What is the hybridization of the central atom in SiCl4? Hybridization = What are the approximate bond angles in this substance? Bond angles = fill in the blank 2° B. What

SOLVED: Hybridization of Si in SiCl4 is sp3d2, sp3d2, sp3d2, sp3d2, sp3d2, sp3d2, sp3d2, sp3d2, sp3d2, sp3d2 and in: SiCl2 is sp3d2
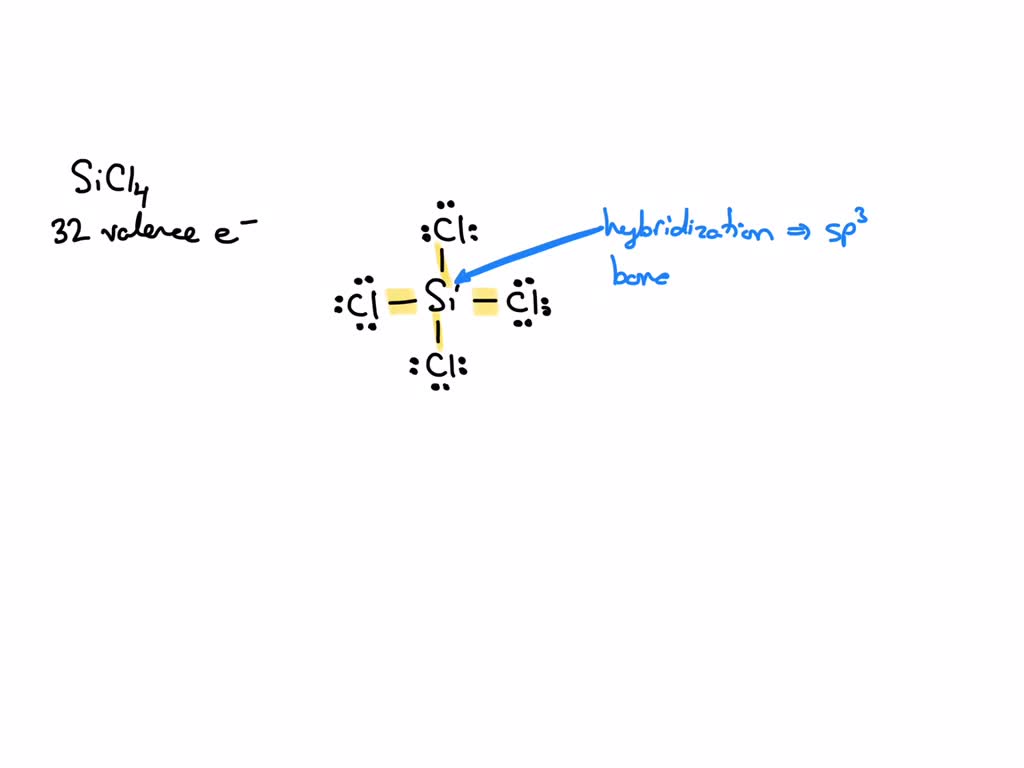
Silicon tetrachloride SiCl4: Molecular Geometry - Hybridization - Molecular Weight - Molecular Formula - Bond Pairs - Lone Pairs - Lewis structure –

SOLVED: 4a) Using Valence Bond Theory, show the hybridization and bonding scheme for silicon tetrachloride (SiCl4): (a) write the atomic orbital diagram for the central atom, (b) circle the atomic orbitals that
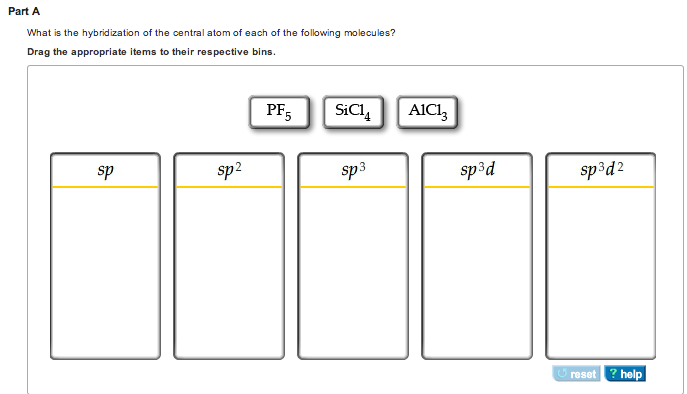
What is the hybridization of the central atom of each of the following molecules? - Home Work Help - Learn CBSE Forum

BCU-NEP-IV SEM-DSC-04-Chemical bonding-Valence Bond Theory(VBT)Sp3 Hybridization-SiCl4 as an Example - YouTube