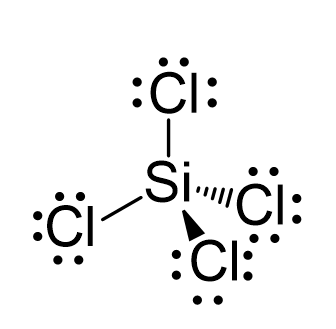
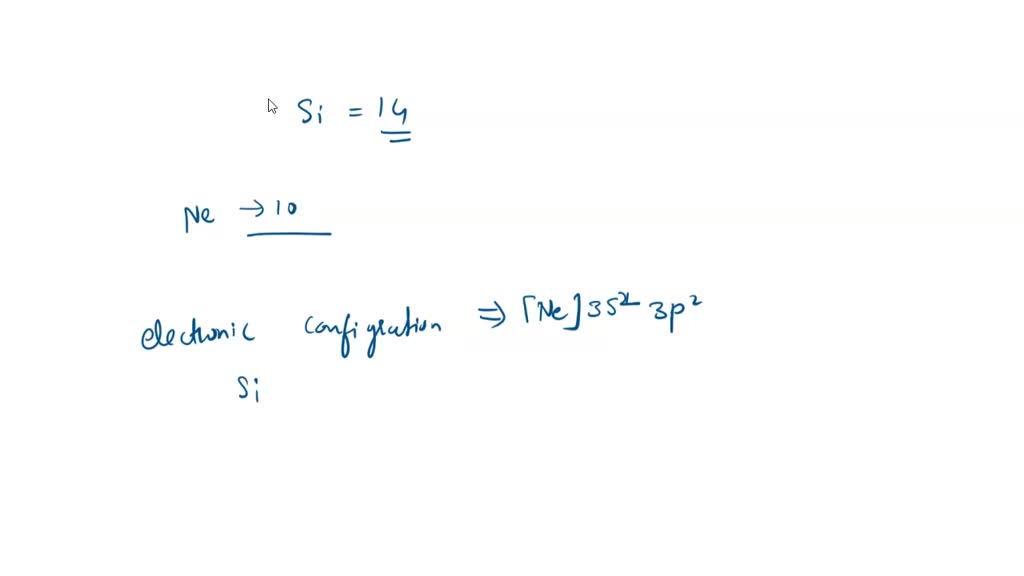Hybridization is a phenomenon that takes place in an atom before chemical bonding. How is hybridization responsible for the observed structure of SiCl4? - Quora

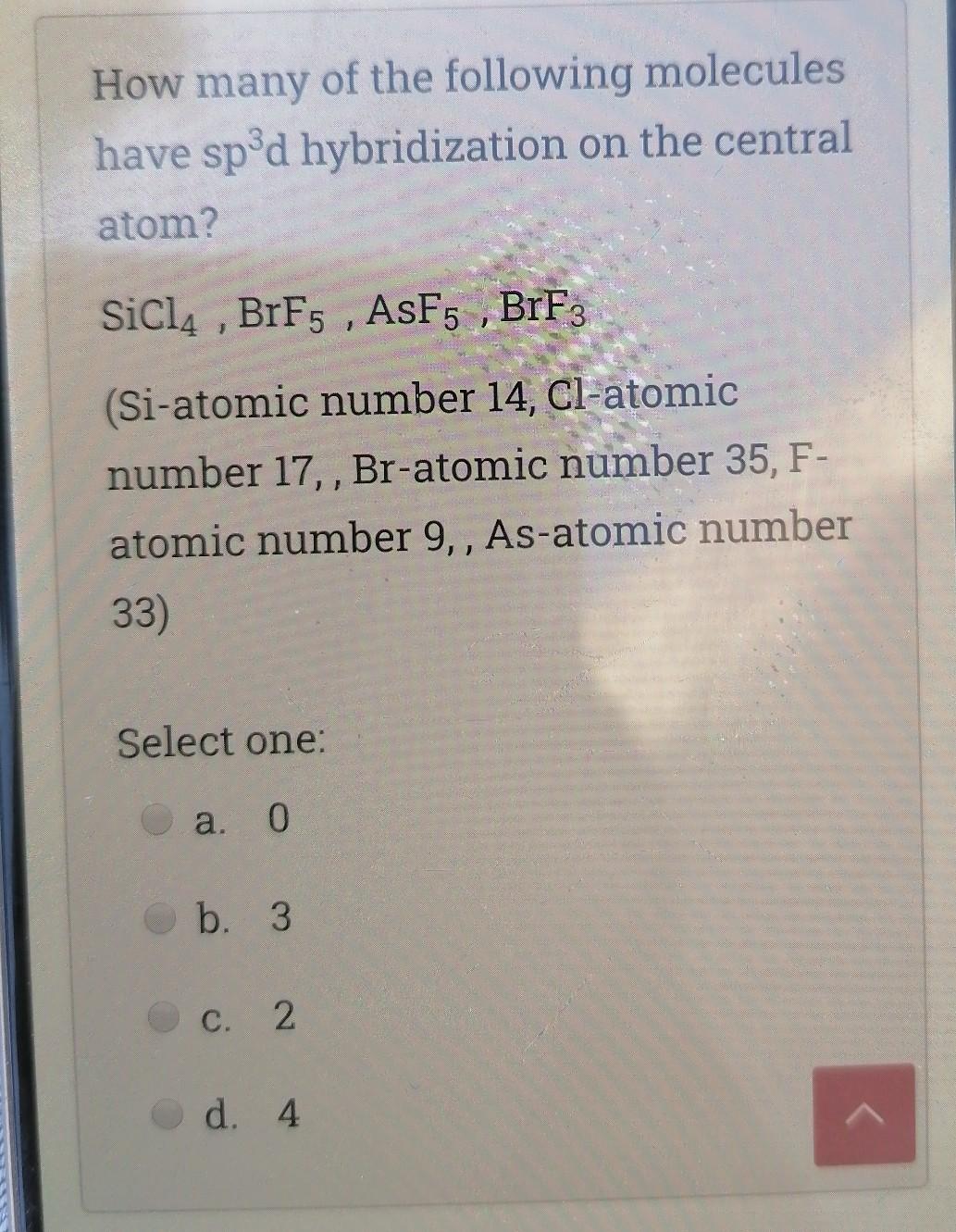
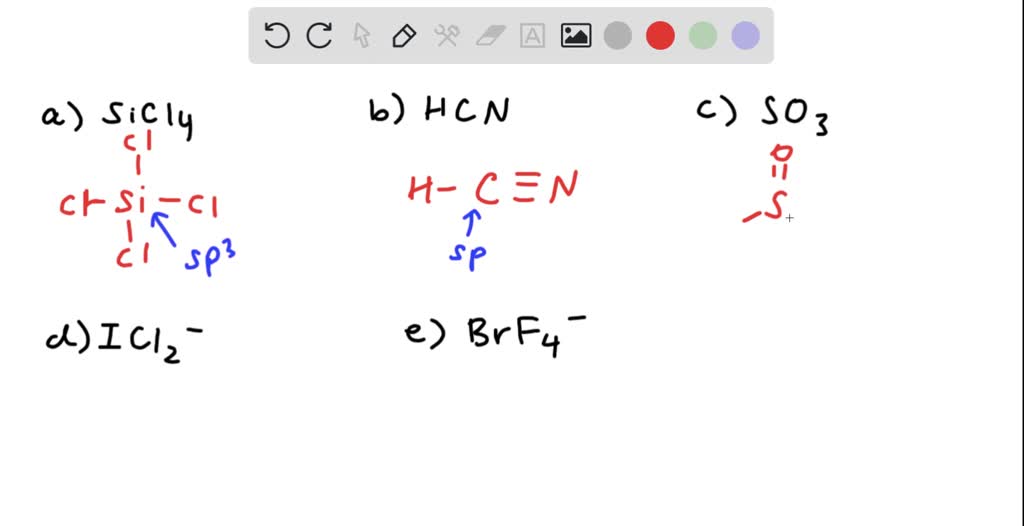
SOLVED: What is the hybridization of the central atom in each of the following: Drag the appropriate items to their respective bins. FeCl2 HCN TeCl2 SiCl4 SO2

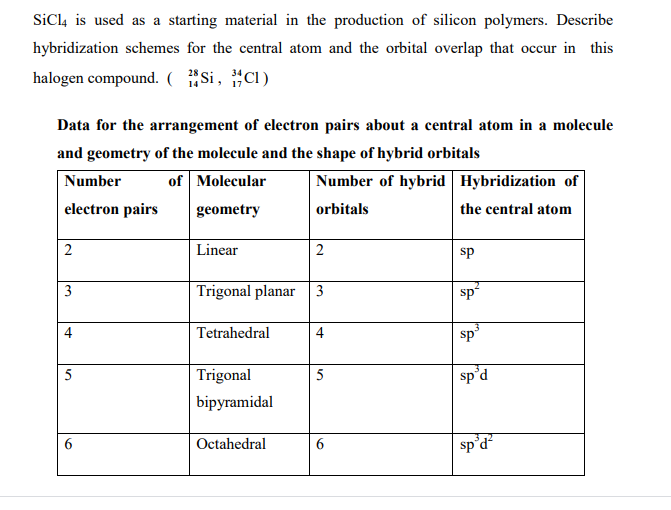
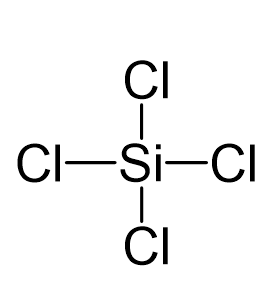
SOLVED: 4a) Using Valence Bond Theory, show the hybridization and bonding scheme for silicon tetrachloride (SiCl4): (a) write the atomic orbital diagram for the central atom, (b) circle the atomic orbitals that