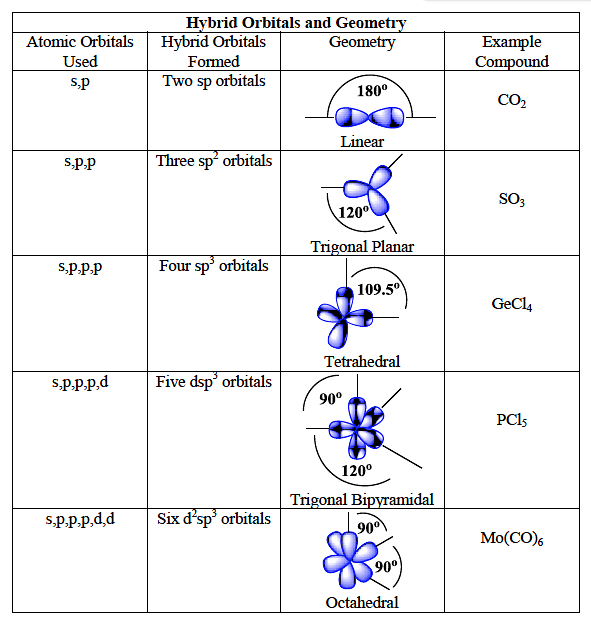
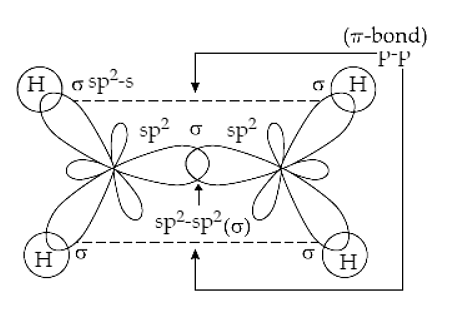
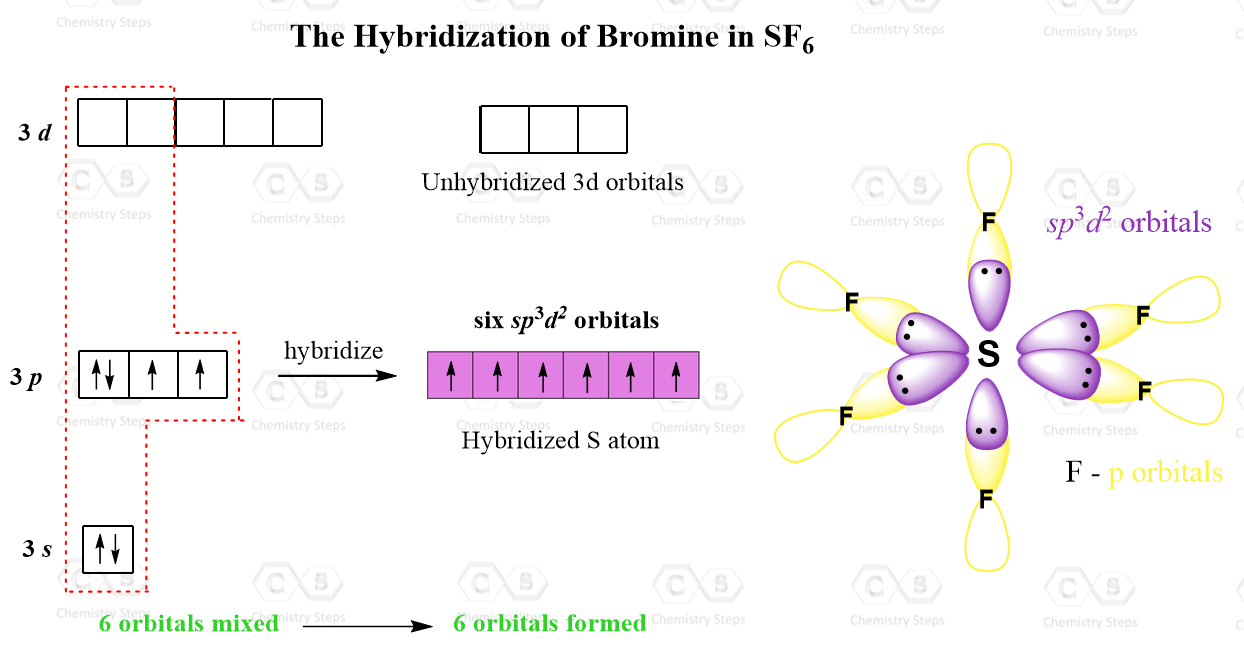
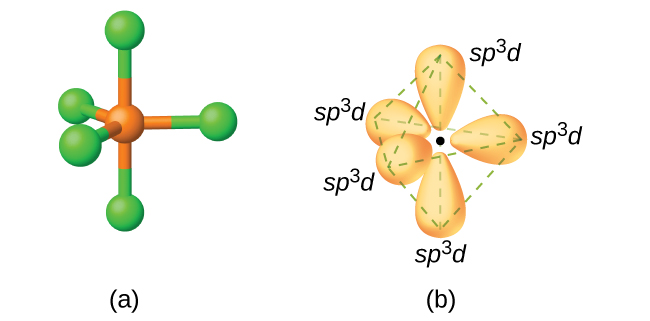
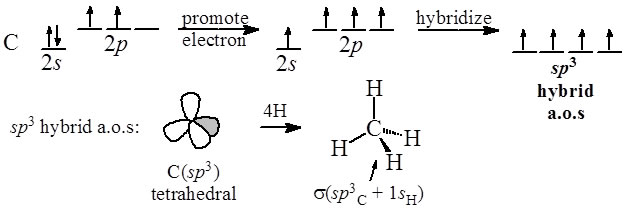
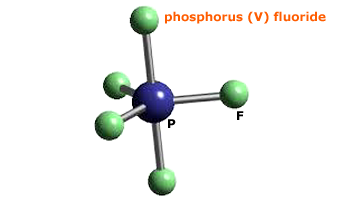
molecular structure - Why an asymmetric geometry with sp3d and sp3d3 hybridization? - Chemistry Stack Exchange

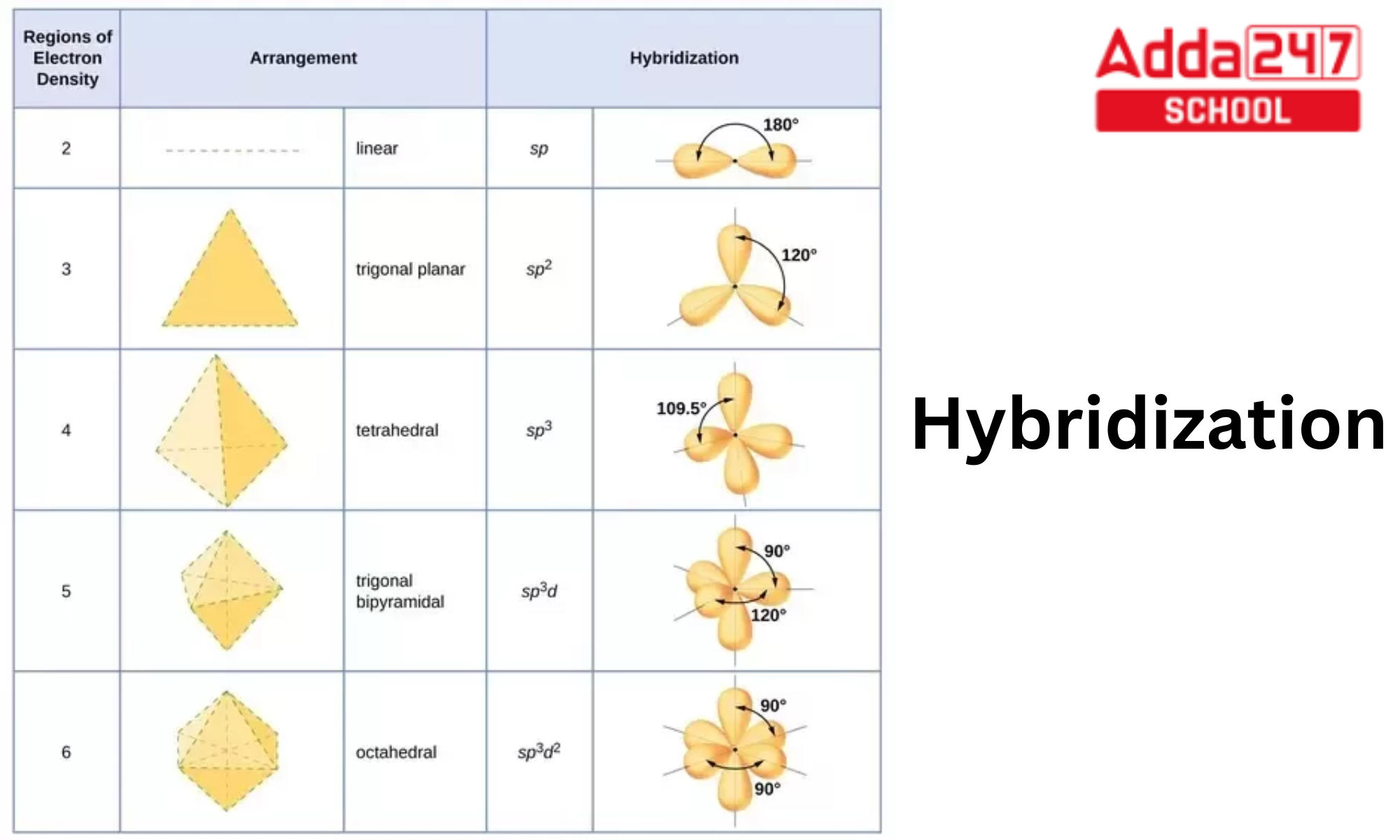
Specify the hybridization of the atoms labeled a-d in the following structure and provide your reasoning for each choice. Your options are sp3, dsp2, sp2, sp3d, or sp. Additionally, provide a brief

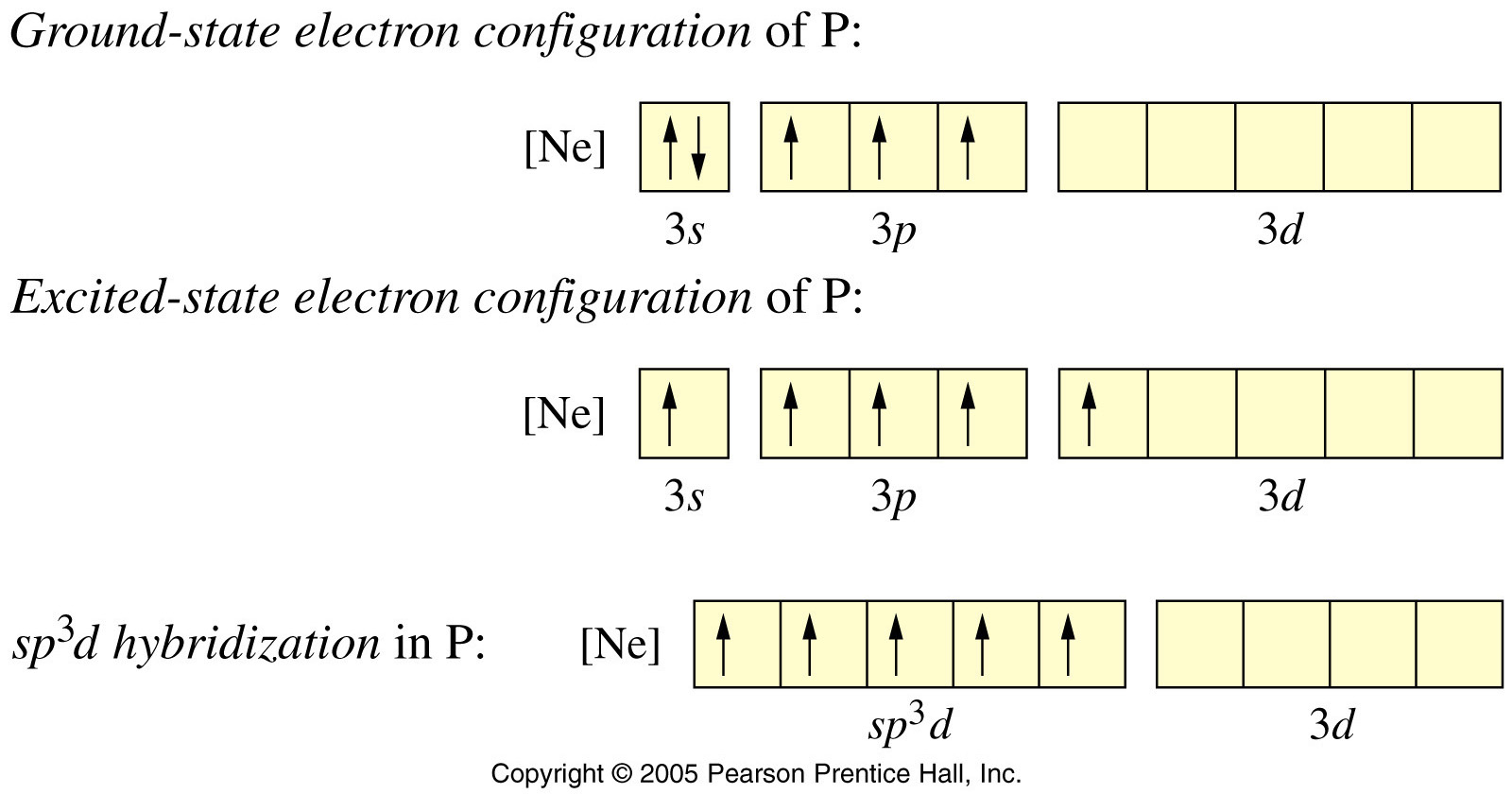
Hybridization || sp3d || sp3d2 || sp3d3 || Formation of PF5, SF6 and IF7 || Chemical Bonding 11th - YouTube
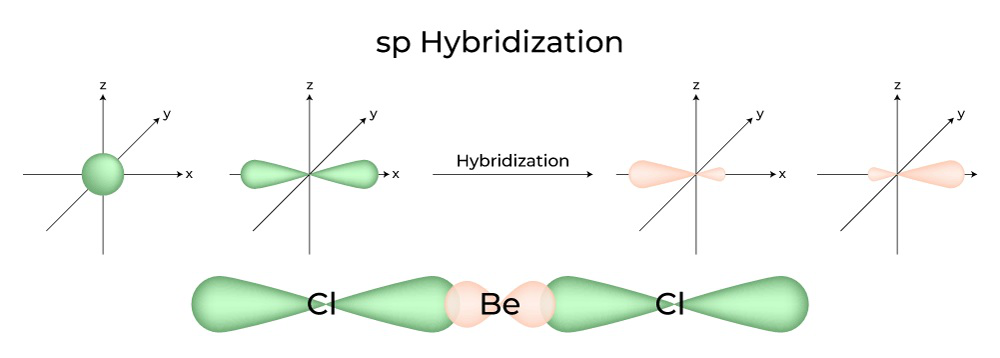
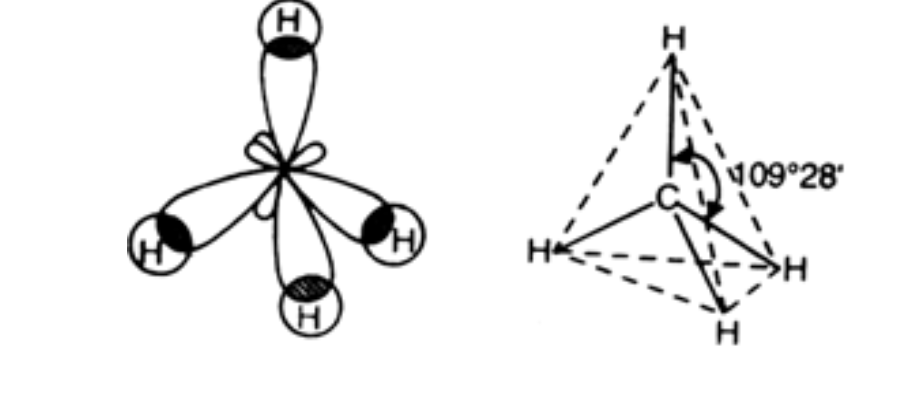
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora
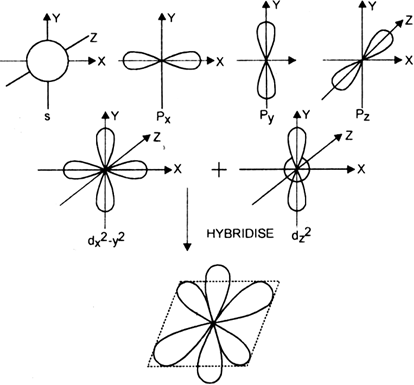
![Explain \\[s{p^3}d\\] hybridization by taking a suitable example. Explain \\[s{p^3}d\\] hybridization by taking a suitable example.](https://www.vedantu.com/question-sets/0801a856-ce79-45dd-96b3-f5dcbf552a724106220300996909947.png)